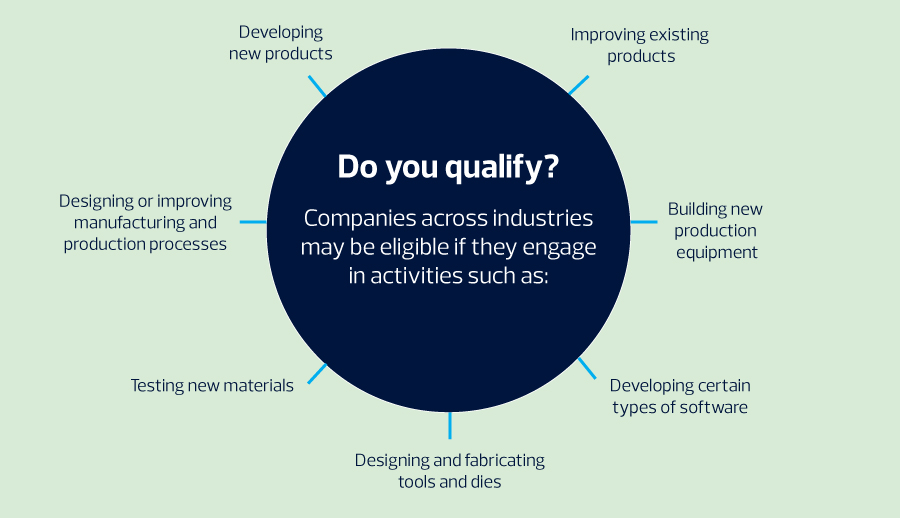
Innovation drives growth—but it’s often expensive. Developing new products, refining processes or investing in emerging technologies requires significant resources. Various federal and state research and development (R&D) tax credits can offset those costs, freeing cash to reinvest in future innovation.
Many middle market companies, however, struggle to identify qualifying activities or document their claims. Rules and requirements can be complex, and sometimes they change. Plus, federal and state rules differ, and many companies lack the time or resources to navigate them effectively. It’s easy to leave valuable credits unclaimed.
RSM’s R&D tax credit professionals work alongside your finance and technical teams to uncover eligible activities, substantiate claims and secure benefits that improve cash flow and reduce your effective tax rate—all while minimizing the disruption to your business.
Why RSM
RSM’s R&D tax credit team combines deep technical knowledge, industry-specific insight and efficient methodologies to help you capture and sustain credits. Our approach focuses on:
- Reducing tax liabilities and increasing cash flow
- Navigating section 174 and section 41 requirements
- Minimizing disruption with sampling and streamlined documentation
- Scaling our process to fit the size and scope of your business
Many middle market businesses do not have the time or resources to gather documentation to withstand IRS or state review. At RSM, we continuously enhance our R&D credit approach to stay aligned with IRS and state requirements and leverage our hands-on experience working with the IRS and state departments to help clients maximize their tax benefits.